
OREM, Utah, Feb. 19, 2026 /PRNewswire/ — Dymicron®, a privately held medical device company advancing next-generation spinal motion-preservation technology, today announced the appointment of Peter Wehrly, one of the spinal industry’s most seasoned C-suite executives, to its Board of Directors. Mr. Wehrly brings more than forty years of leadership experience, including roles as CEO, President, Chairman, and global […]

Cervical total disc replacement (TDR) continues to gain traction globally as a motion-preserving alternative to spinal fusion for appropriately selected patients. Recently, a clinical milestone was achieved with the first cervical total disc replacement surgery performed in Paraguay, utilizing the Triadyme®-C cervical artificial disc. The procedure was completed at the Centro Médico La Costa in Asunción, Paraguay, by Elio […]

Dymicron®, a privately held medical device company pioneering advanced spinal technologies, today announced the successful enrollment and treatment of the first patient in its Triadyme®-C Investigational Device Exemption (IDE) clinical trial. This milestone marks the initiation of a U.S. clinical study evaluating the safety and effectiveness of the Triadyme-C cervical artificial disc.
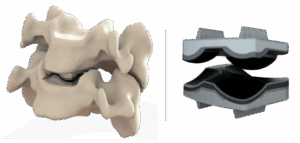
Dymicron®, a privately held medical device company pioneering advanced spinal technologies, today announced that the United States (U.S.) Food and Drug Administration (FDA) has granted Investigational Device Exemption (IDE) approval to begin a pivotal clinical study of the Triadyme®‑C cervical artificial disc. This achievement marks a critical milestone in the company’s mission to bring the […]

Dymicron®, a pioneering medical technology company renowned for its revolutionary cervical spine disc replacement solution, has announced the appointment of Dominik Wilke as its new Senior Manager, Business Development and Marketing. With over a decade of experience in the spinal device industry, Dominik brings a wealth of expertise and a passion for innovation to his new […]

Dymicron® is proud to be recognized in the annual listing of 10 companies at the forefront of biomechanics solutions.

Dymicron’s Triadyme-C Artificial Cervical Disc won the 2021 Best New Spine Technology Award presented by Orthopedics This Week.

Dymicron has developed a next-generation man-made diamond material for orthopedic applications in its Triadyme®-C artificial cervical disc replacement.
Dymicron appoints Gunther Peeters as Senior Vice President of Commercial Operations to lead international market expansion.
Dr. Khachatryan joins Dymicron’s Board of Directors, bringing extensive experience in cervical arthroplasty.
Sign up to discover the latest news about Dymicron and our proprietary polycrystalline diamond technology–Adymite. Adymite is redefining performance and reliability in spinal disc replacement, including delivering unmatched durability and excellent long-term outcomes for patients.